With concerns swirling around the efficacy and safety of each COVID-19 vaccine, the New England Journal of Medicine published a peer-reviewed report confirming that “the vaccine known as mRNA-1273 was 94.1% efficacious in preventing symptomatic coronavirus disease 2019 (COVID-19).” The Moderna CovID-19 vaccine also demonstrated efficacy in preventing severe versions of the illness. Investigators identified no safety concerns and no evidence of vaccine-associated enhanced respiratory disease (VAERD).
According to the authors of the report:
There were no concerning safety issues with vaccination, according to the authors. Local reactions to the vaccine were generally mild. About 50% of participants receiving mRNA-1273 experienced moderate-to-severe side effects–such as fatigue, muscle aches, joint pain and headache–after the second dose, which resolved in most volunteers within two days.
Investigators also observed no evidence of VAERD among those who received mRNA-1273. This rare complication was seen in individuals vaccinated with a whole-inactivated respiratory syncytial virus (RSV) vaccine in the 1960s, before there was a capacity to define protein structures and measure immune responses with precision. VAERD can occur when a vaccine induces an immune response that is not strong enough to protect against infection.
The study resulted in no concerning safety issues with vaccination, according to the authors of the report. Local reactions to the vaccine were generally mild. About 50% of participants receiving mRNA-1273 experienced moderate-to-severe side effects—such as fatigue, muscle aches, joint pain and headache—after the second dose, which resolved in most volunteers within two days. The trial was led by principal investigators Lindsey R. Baden, M.D. of Brigham and Women’s Hospital in Boston, Hana M. El-Sahly, M.D. of Baylor College of Medicine in Houston, and Brandon Essink, M.D., of Meridian Clinical Research.
RELATED: DO UNIVERSITIES NEED VACCINE MANDATES?
This information was released by NIH/National Institute of Allergy and Infectious Diseases, published in the New England Journal of Medicine, and funded by the National Institute of Allergy and Infectious Diseases.
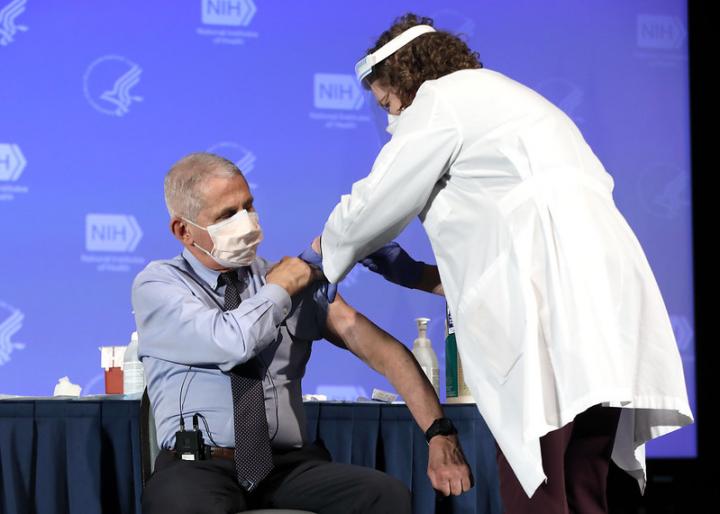
Image: Dr. Anthony Fauci, Director of the National Institute of Allergy and Infectious Diseases, received the Moderna COVID-19 vaccine at the HHS/NIH COVID-19 Vaccine Kick-Off event at NIH on 12/22/20. (NIH)
The information contained in this article is for informational purposes only and is not intended as health or medical advice. Always consult a physician or other qualified health provider regarding any questions you may have about a medical condition or health objectives.